Sodium Nitrate Crystal holds significant potential across various industries, from agriculture to food preservation. Experts estimate that the global market for sodium nitrate will reach approximately $1.4 billion by 2025. This growth reflects increased demand in agriculture, where it serves as a vital fertilizer source. Notably, Dr. Emily Gray, a leading expert in agricultural chemistry, states, "Using Sodium Nitrate Crystal effectively can significantly enhance crop yield and quality."
In crafting projects that utilize Sodium Nitrate Crystal, it is essential to appreciate its properties. For instance, this compound acts not only as a powerful fertilizer but also as a preservative in food processing. However, improper usage can lead to unintended negative consequences, such as soil depletion over time. Awareness of these concerns can help mitigate errors in application, enhancing overall project outcomes.
Furthermore, many are unaware of the fine balance required when applying Sodium Nitrate Crystal. Results can vary based on soil composition and climate. As one dives deeper into the practical applications of Sodium Nitrate Crystal, recognizing these nuances will prove crucial for success. It's a complex yet rewarding journey in understanding how best to integrate this compound into diverse projects.

Sodium nitrate is a versatile compound with significant applications across various industries. Its chemical formula, NaNO3, indicates a strong ionic bond structure. This compound acts as an oxidizing agent in chemical reactions. In agriculture, it serves as a nitrogen fertilizer. Studies show that it can improve crop yields by 20-30% under optimal conditions. However, over-reliance on sodium nitrate raises concerns about soil salinity. Balancing its use is crucial for sustainable farming.
In food preservation, sodium nitrate plays a key role. It inhibits bacterial growth in cured meats, extending shelf life. The World Health Organization highlights the importance of controlling sodium nitrate levels in food. Excessive intake can lead to health risks. Many professionals advocate for clear labeling and mindful consumption practices.
Despite its benefits, sodium nitrate is not without risks. Inappropriate usage can lead to environmental contamination. This can affect water sources, harming aquatic life. Monitoring and strict regulations are essential to mitigate these risks. Understanding the properties and ethical applications of sodium nitrate is pivotal for responsible project execution. Careful consideration can enhance its positive impact while minimizing harm.
| Property | Value | Application |
|---|---|---|
| Chemical Formula | NaNO3 | Fertilizers |
| Molar Mass | 85.00 g/mol | Food Preservation |
| Appearance | White crystalline solid | Chemical Manufacturing |
| Solubility | Soluble in water | Heat Transfer Agents |
| Melting Point | 308 °C | Explosives |
| Boiling Point | 380 °C | Antifreeze Solutions |
| pH Level | 6.0 - 7.0 | Agricultural Applications |

When handling sodium nitrate crystals, safety should be your top priority. According to a recent report by the International Chemical Safety Card, sodium nitrate can cause irritation to skin and eyes. Always wear appropriate personal protective equipment (PPE) like gloves and goggles. This protective gear minimizes exposure risks, acting as a first line of defense.
Proper ventilation is crucial. Inhalation of sodium nitrate dust can lead to respiratory issues. Ensure that you work in a well-ventilated area. If working indoors, consider using exhaust fans or air purifiers. The Occupational Safety and Health Administration recommends exposure limits of 10 mg/m³ for dust. Adhering to this limit reduces health risks significantly.
Store sodium nitrate in a cool, dry place. Containers should be tightly sealed to prevent moisture absorption. Moisture can lead to clumping, making the substance hard to use. Regularly inspect storage conditions to ensure safety and effectiveness. These practices can help avoid unintended chemical reactions, which are particularly hazardous. The unpredictable nature of chemicals can lead to regrettable situations if safety is overlooked.
To dissolve sodium nitrate effectively, choose the right solvent. Water is the most common choice, but others can work too. Warm water enhances solubility. It’s important to measure the water accurately. Too little water can lead to incomplete dissolution.
When adding sodium nitrate to water, stir continuously. This helps the crystals break down quickly. Opt for a beaker instead of a flask. Beakers provide more space for mixing. After a few minutes, check for undissolved crystals. Sometimes, even with stirring, it won't fully dissolve. This can be frustrating but informative.
If you're using ethanol, the approach differs. Ethanol is less effective, so larger quantities might be necessary. The process takes longer, and patience is key. Still, it's valuable to experiment and learn from these attempts. Every project offers room for improvement.
Sodium nitrate can be a fascinating addition to various DIY projects. Understanding how to use it effectively is key. This compound can enhance soil for gardening or even create vibrant colors in educational experiments. It's powerful.
When integrating sodium nitrate, consider its solubility in water. Dissolving the crystals can improve nutrient absorption in plants. For instance, adding it to watering solutions can nurture your garden. Just be careful; too much can harm plants. Watch the concentration closely.
Tips: Start with small amounts. Always test on a few plants. Monitor their growth. This experimentation can help you discover what works best. Remember, each plant might react differently.
For creative projects, sodium nitrate can also be mixed with other substances for visual effects. Think about school science projects that demonstrate chemical reactions. However, safety is crucial. Always wear gloves and eye protection. Mistakes can lead to unexpected results. Reflect on your process and adjust as needed.
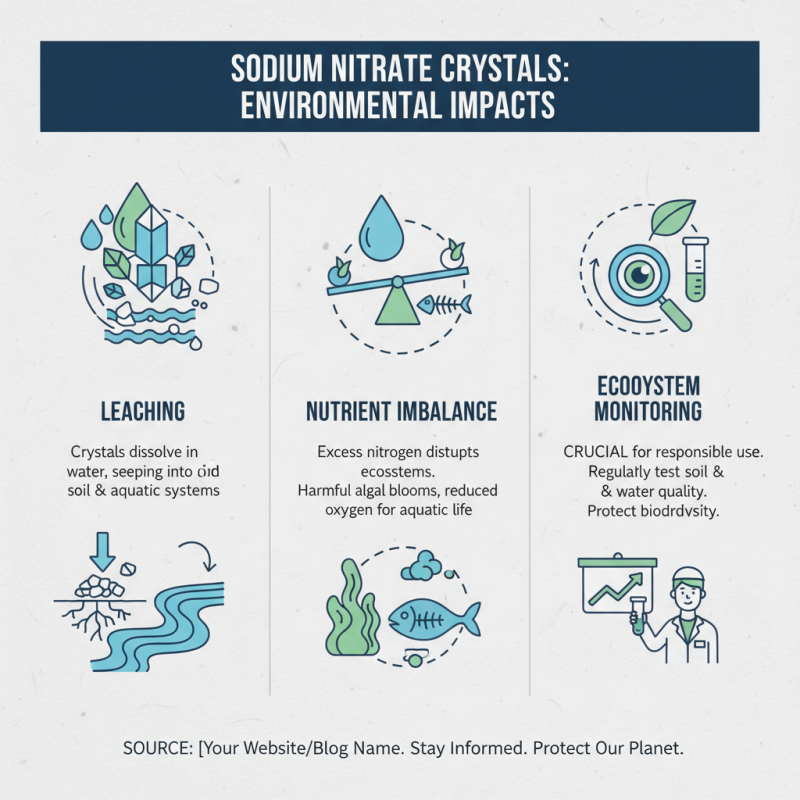
Using sodium nitrate crystals can have significant environmental impacts. These crystals may leach into the soil and water during use. This process can lead to nutrient imbalance in ecosystems. Monitoring local ecosystems is essential when employing this chemical in your projects.
Proper storage is also important. Ensure that sodium nitrate crystals are kept in a dry, cool place. Leakage from improper containers can occur. While using these crystals, be cautious about runoff during rain. It can carry harmful substances to nearby water sources. Awareness of local wildlife is critical as well. They may be affected by altered soil and water quality.
Reflecting on the application methods is necessary. Are there safer alternatives available? Often, rethinking our approach can lead to better outcomes. Engage with local environmental guidelines. These resources can provide valuable insights into safe usage. Balancing project needs with environmental health is a continual challenge.