Industrial Sodium Nitrite plays a crucial role in various sectors, including food preservation and chemical manufacturing. Dr. Emily Johnson, a leading expert in industrial chemicals, states, “Industrial Sodium Nitrite is essential for maintaining product safety and quality.” This compound not only acts as a preservative but also serves as a versatile intermediate in chemical syntheses.
In food processing, Industrial Sodium Nitrite prevents bacterial growth and enhances color. It is especially important in meat products. However, its use requires careful regulation to avoid health risks. As industries rely on this compound, it's vital to address safety standards and potential risks.
Understanding the applications and implications of Industrial Sodium Nitrite can lead to better practices. This knowledge is not only for manufacturers but also for consumers. Balancing benefits with safety demands ongoing reflection and adaptation in this field.

Industrial sodium nitrite is a versatile inorganic compound with key applications across various industries. It is primarily recognized for its role as a preservative in the food industry, particularly in curing meats. The compound appears as a white crystalline powder and has a chemical formula of NaNO2. One of its most notable characteristics is its ability to inhibit the growth of harmful bacteria, particularly Clostridium botulinum. According to the US Food and Drug Administration (FDA), sodium nitrite is allowed in certain quantities, showcasing its recognized safety in specified applications.
In addition to food preservation, industrial sodium nitrite serves critical roles in the chemical and pharmaceutical industries. It acts as a dye intermediate and a corrosion inhibitor. The market for sodium nitrite is projected to grow significantly. A recent report by MarketResearchFuture estimates a compound annual growth rate (CAGR) of around 5% from 2021 to 2026. This growth is fueled by increasing industrial demand for nitrites in various applications. However, users must be aware of the compound's environmental impact. Improper disposal can lead to pollution, prompting discussions about sustainable practices in sodium nitrite production and use.
The characteristics of sodium nitrite also raise safety considerations. While it is generally safe in controlled environments, exposure to high concentrations can be hazardous. This underscores the importance of strict regulations and handling procedures in industries that utilize sodium nitrite. Balancing its benefits with potential risks remains a central concern among professionals in the field.
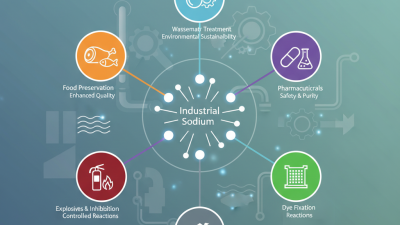
Industrial sodium nitrite finds broad applications across various sectors. In the food industry, it serves as a curing agent. Meat processors rely on it to enhance flavor and preserve color in products like ham and bacon. Sodium nitrite inhibits bacterial growth, particularly against Clostridium botulinum, making cured meats safer for consumption.
In the realm of pharmaceuticals, sodium nitrite is employed as a reagent in the synthesis of numerous compounds. It plays a role in developing medications for conditions such as hypertension. Furthermore, in the dye industry, it acts as an intermediate for dye production, contributing to a wide range of colorants used in textiles.
However, the use of sodium nitrite comes with caution. Exposure can lead to health risks. Therefore, proper guidelines and safety measures are critical. Industries must prioritize monitoring and ensure adequate training for workers. It's essential to balance its benefits with the potential risks associated with misuse or overexposure.
| Industry | Application | Benefits |
|---|---|---|
| Food Processing | Preservative for meats | Inhibits bacteria growth, enhances flavor and color |
| Textiles | Dyeing and finishing agent | Improves color stability and brightness |
| Chemical Manufacturing | Production of other chemicals | Essential intermediate for many reactions |
| Pharmaceuticals | Used in drug formulations | Source of nitrogen for various compounds |
| Water Treatment | Corrosion inhibitor | Protects metal surfaces in systems |

Sodium nitrite is a chemical used widely in various industries, especially in food preservation and pharmaceuticals. However, handling it requires careful consideration of health and safety. Sodium nitrite can be hazardous when inhaled or ingested in large amounts. Exposure may lead to headaches, nausea, and in severe cases, methemoglobinemia, a condition that affects the blood’s ability to carry oxygen.
When working with sodium nitrite, proper personal protective equipment (PPE) is essential. Gloves, goggles, and masks can help reduce exposure risks. The OSHA standards recommend monitoring air levels for safe working conditions, particularly in confined spaces. Industry reports indicate that proper ventilation can significantly lower inhalation risks. It is also crucial to store sodium nitrite securely, away from incompatible substances.
Tips: Always read the safety data sheet (SDS) before using sodium nitrite. Ensure your workspace is well-ventilated and avoid direct contact. Think twice before handling chemicals; awareness of risks can lead to safer practices. Regular training is vital to reinforce safety protocols among workers. Implementing these measures can help mitigate risks and promote a safer environment.
Sodium nitrite is widely used in various industries, particularly in food preservation and pharmaceuticals. However, its environmental impact and regulatory standards are significant concerns. Sodium nitrite can contaminate soil and water. When released into the environment, it can form nitrosamines, which are harmful compounds.
Regulatory bodies set strict limits on sodium nitrite levels in food and industrial applications. The United States Environmental Protection Agency (EPA) establishes limits for wastewater discharge. Many countries have similar guidelines to protect public health. These regulations are crucial for minimizing risks associated with sodium nitrite exposure.
Awareness is growing regarding the potential risks of sodium nitrite. Industries must adapt their practices to adhere to these standards. It's essential to find safer alternatives where possible. Sustainable practices can help minimize the environmental footprint of sodium nitrite production and usage. Balancing industrial needs with safety and environmental considerations requires continuous effort and reflection.
Sodium nitrite is gaining attention for its diverse applications in various industries. Recent trends highlight a shift towards more sustainable practices. Innovations focus on optimizing sodium nitrite's usage, particularly in food preservation and pharmaceuticals.
Research has explored using sodium nitrite in modified atmospheres for packaging. This technique extends shelf life while maintaining food quality. Emerging studies suggest sodium nitrite may play a role in innovative therapies, particularly in treating specific medical conditions. It is essential to remain cautious with these developments, ensuring safety and efficacy.
Tips: Always consult with professionals before using sodium nitrite for any application. Monitor new research to stay updated on safe practices. Experimentation could lead to unexpected results, so approach with care.