Concentrated Nitric Acid plays a crucial role in various industries. Dr. James Carter, an expert in chemical engineering, once stated, "Concentrated Nitric Acid is a driving force in modern manufacturing." This powerful acid is essential in producing fertilizers, explosives, and pharmaceuticals.
Its applications extend to metal processing, where it is crucial for etching and cleaning metals. In the world of science, it aids in chemical synthesis and analysis, helping researchers obtain vital data. Despite its valuable uses, working with concentrated nitric acid requires cautious handling due to its corrosive nature.
Inefficiencies can arise if proper safety protocols are not followed. The industry continuously seeks ways to improve processes. Reflecting on experiences can lead to advancements in safety and efficiency with concentrated nitric acid. By embracing technology and innovation, industries can enhance their productivity while prioritizing safety.

Concentrated nitric acid plays a crucial role in fertilizer production. It is primarily used to synthesize ammonium nitrate. This compound is vital for agriculture. Farmers rely on ammonium nitrate to boost crop yields. The acid reacts with ammonia to produce this essential fertilizer.
Tips: Always handle concentrated nitric acid with care. Use proper safety gear like gloves and goggles. Ventilation is important.
Another application is in the production of nitrate fertilizers. This includes various formulations tailored for specific crops. The right balance of nutrients can enhance plant growth. While effective, creating these fertilizers is complex. It requires precise measurements and processes. Mistakes can lead to ineffective products or environmental harm.
Tips: Regularly inspect equipment to avoid leaks. Consistency in measurements ensures quality. Always review safety protocols to protect the environment.
Concentrated nitric acid plays a crucial role in the manufacture of explosives. It acts as an oxidizer, facilitating chemical reactions. This highly reactive substance can create a variety of explosive compounds, like nitroglycerin and TNT. These materials are essential for both military and industrial applications.
In processing, concentrated nitric acid must be handled with extreme care. Its corrosive nature can damage equipment and pose safety hazards. Workers need proper training to manage its risks. Even minor spills can lead to significant consequences. The industry must constantly evaluate safety protocols to prevent accidents.
The production of explosives using concentrated nitric acid raises ethical concerns. The potential for misuse is significant. Society must reflect on how these materials are used. Balancing scientific advancement with safety remains a complex issue. These discussions are vital as we navigate the challenges of using such powerful chemicals.
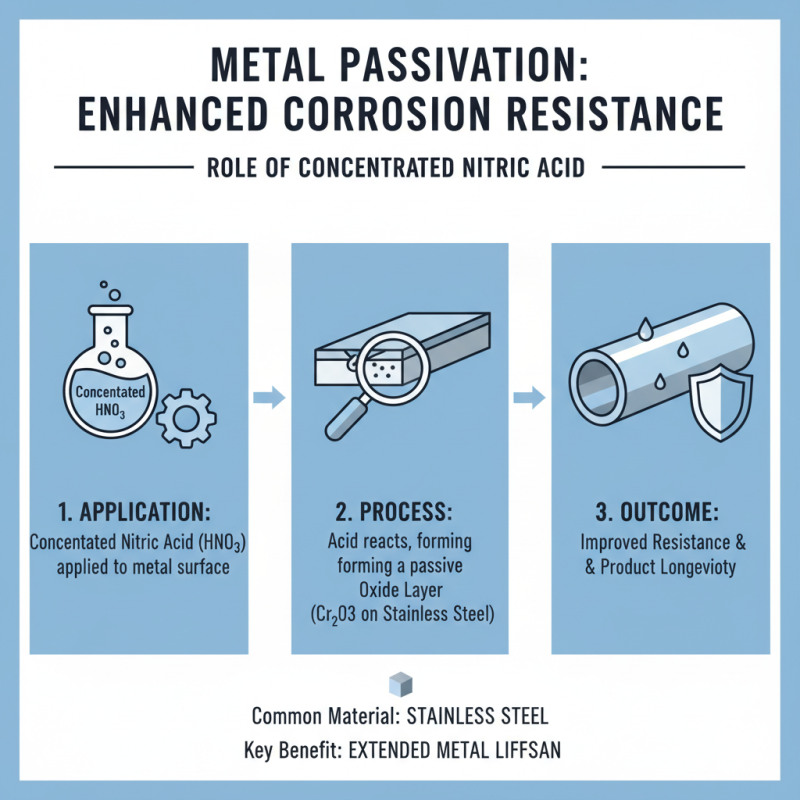
Concentrated nitric acid plays a crucial role in metal passivation processes. Passivation enhances the corrosion resistance of metals. This chemical forms a protective oxide layer on the surface. Stainless steel is a common material subjected to this process. The application of nitric acid can significantly improve the longevity of metal products.
When applying concentrated nitric acid, careful monitoring is essential. The acid must be used in a controlled environment to avoid excessive reaction rates. Too much exposure can damage the underlying metal, negating the benefits of passivation. Workers need proper training when handling this chemical.
Ventilation is vital during the process. Fumes from concentrated nitric acid can be hazardous. Protective equipment should always be worn. Even small spills require immediate attention. Reflecting on safety and efficiency is key. Balancing efficacy with safety standards can be challenging. Continuous improvement in these practices is necessary for industry advancement.
Concentrated nitric acid plays a vital role in laboratory synthesis reactions. Its strong oxidizing properties enable chemists to create various compounds. Common uses include nitration processes, which are essential in producing explosives and pharmaceuticals. This versatility makes it a preferred reagent in many reactions.
The acid's ability to act as a dehydrating agent is another reason it is significant in laboratories. It removes water from reactions, enhancing yields. However, handling concentrated nitric acid requires careful consideration. It is highly corrosive and can react unpredictably with other substances. Proper safety measures are crucial to avoid accidents.
While concentrated nitric acid offers various benefits, it poses challenges too. Its strong fumes can be harmful if inhaled. Working in a well-ventilated environment is advisable. Researchers must balance its applications and risks thoughtfully. Mistakes can lead to unfortunate outcomes, underscoring the need for diligence in its use.
| Application Area | Description | Significance | Key Reactions/Involvements |
|---|---|---|---|
| Fertilizer Production | Used in the synthesis of ammonium nitrate. | Essential for agricultural fertilizers providing nitrogen. | NH₃ + HNO₃ → NH₄NO₃ |
| Explosives Manufacture | Key component in TNT and other explosive compounds. | Crucial for military and industrial applications. | C₇H₅N₃O₆ + HNO₃ → TNT |
| Metal Etching | Used for etching metals like copper and aluminum. | Important in electronics and manufacturing. | Cu + 4HNO₃ → Cu(NO₃)₂ + 2NO₂ + 2H₂O |
| Nitric Acid in Organic Synthesis | Used to produce nitro compounds from hydrocarbons. | Significant in pharmaceuticals and dye industries. | C₆H₆ + HNO₃ → C₆H₅NO₂ + H₂O |
| Water Treatment | Utilized for the removal of heavy metals in industrial wastewater. | Essential for environmental protection. | Heavy metals + HNO₃ → Complex ions |
| Chemical Analysis | Commonly used in titrations and qualitative analysis. | Essential for laboratory research and industrial quality control. | Various compounds + HNO₃ → Products |
| Production of Nitrates | Used to synthesize various nitrates. | Important for fertilizers and chemical feedstock. | Metal oxides + HNO₃ → Metal nitrates |
| Laboratory Reagent | Used as a strong oxidizing agent in synthesis. | Fundamental in research institutions. | Multiple organic reactions. |
| Density Measurement | Used in precision density measurements of liquids. | Important in quality assurance processes. | Determining specific gravity. |
| Nitric Oxide Production | Formed during the combustion processes involving nitrogen. | Significant in environmental studies and NOx emissions. | N₂ + O₂ → 2NO |

Concentrated nitric acid plays a crucial role in environmental testing and analysis. This chemical compound is widely used for its ability to oxidize organic materials. In water quality testing, nitric acid is essential for digesting samples. It helps release metals and other contaminants, allowing for accurate analysis. Reports indicate that about 70% of labs utilize nitric acid for such purposes.
In soil analysis, concentrated nitric acid aids in nutrient determination. It helps extract elements like phosphorus and potassium efficiently. Data from environmental studies show that over 60% of soil testing labs rely on this method. However, it's noteworthy that handling nitric acid requires strict safety measures. Exposure can lead to serious health risks if not managed properly.
While nitric acid has proven benefits, its environmental impact raises concerns. Improper disposal can lead to water contamination. In some studies, nitric acid residue has been detected in surrounding ecosystems. This highlights the necessity for better waste management practices in industries using nitric acid. Balancing its advantages in testing with its potential harm is an ongoing challenge in the field.